 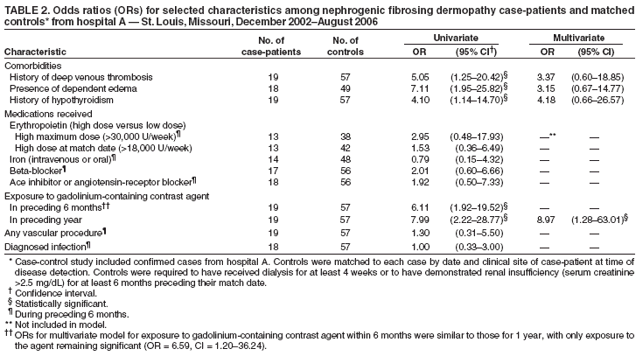
1 - 4 After more than 500 cases of NSF were reported from 1997 to 2007, regulations were adopted to prevent NSF. Nephrogenic systemic fibrosis (NSF) is a rare, potentially fatal condition caused by iatrogenic gadolinium administration in patients with acute kidney injury or stage 4 or 5 chronic kidney disease (CKD), defined as an estimated glomerular filtration rate (eGFR) of less than 30 mL/min/1.73 m 2. Trial Registration PROSPERO identifier: CRD42019123284 The potential diagnostic harms of withholding group II GBCA for indicated examinations may outweigh the risk of NSF in this population. The upper bound varied owing to different sample sizes for gadobenate dimeglumine (0 of 3167 upper bound of 95% CI, 0.12%), gadoterate meglumine (0 of 1204 upper bound of 95% CI, 0.31%), gadobutrol (0 of 330 upper bound of 95% CI, 1.11%), and gadoteridol (0 of 230 upper bound of 95% CI, 1.59%).Ĭonclusions and Relevance This study’s findings suggest that the risk of NSF from group II GBCA administration in stage 4 or 5 CKD is likely less than 0.07%. The pooled incidence of NSF was 0 of 4931 (0% upper bound of 95% CI, 0.07%). 
Results Sixteen unique studies with 4931 patients were included (κ = 0.68) in this systematic review and meta-analysis. Main Outcomes and Measures Pooled incidence of NSF and the associated upper bound of a 2-sided 95% CI (risk estimate) for the pooled data and each of the 4 group II GBCAs. Conference abstracts, retracted manuscripts, narrative reviews, editorials, case reports, and manuscripts not reporting total group II GBCA administrations were excluded.ĭata Extraction and Synthesis Data extraction was performed for all studies by a single investigator, including publication details, study design and time frame, patient characteristics, group II GBCA(s) administered, total exposures for patients with stage 4 or stage 5 CKD, total cases of unconfounded NSF, reason for GBCA administration, follow-up duration, loss to follow-up, basis for NSF screening, and diagnosis. Inclusion criteria consisted of stage 4 or 5 CKD with or without dialysis, administration of an unconfounded American College of Radiology classification group II GBCA (gadobenate dimeglumine, gadobutrol, gadoterate meglumine, or gadoteridol), and incident NSF as an outcome. Agreement for final cohort inclusion was determined by 2 blinded screeners using Cohen κ. Study Selection Citations were screened for inclusion in a multistep process. Objective To assess the pooled risk of NSF in patients with stage 4 or 5 CKD receiving a group II GBCA.ĭata Sources A health sciences informationist searched the Ovid (MEDLINE and MEDLINE Epub Ahead of Print, In-Process & Other Non-Indexed Citation, and Daily and Versions), Embase, Cochrane Central Register of Controlled Trials, Web of Science, and Open Grey databases from inception to January 29, 2019, yielding 2700 citations. Importance Risk of nephrogenic systemic fibrosis (NSF) to individual patients with stage 4 or 5 chronic kidney disease (CKD defined as estimated glomerular filtration rate of <30 mL/min/1.73 m 2) who receive a group II gadolinium-based contrast agent (GBCA) is not well understood or summarized in the literature. Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
